Reasons for slowing down the process of Covid-19 Pandemic
The global number of COVID-19 cases and deaths has fallen for the sixth week in a row, marking a sharp downward trend in the pandemic. So much so, in fact, that many countries are planning a return to something resembling “normality” by June or July.

The FDA has authorized emergency use for the Johnson & Johnson vaccine
The U.S. Food and Drug Administration issued an emergency use authorization (EUA) for the third vaccine for the prevention of coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The EUA allows the Janssen COVID-19 Vaccine to be distributed in the U.S for use in individuals 18 years of age and older.
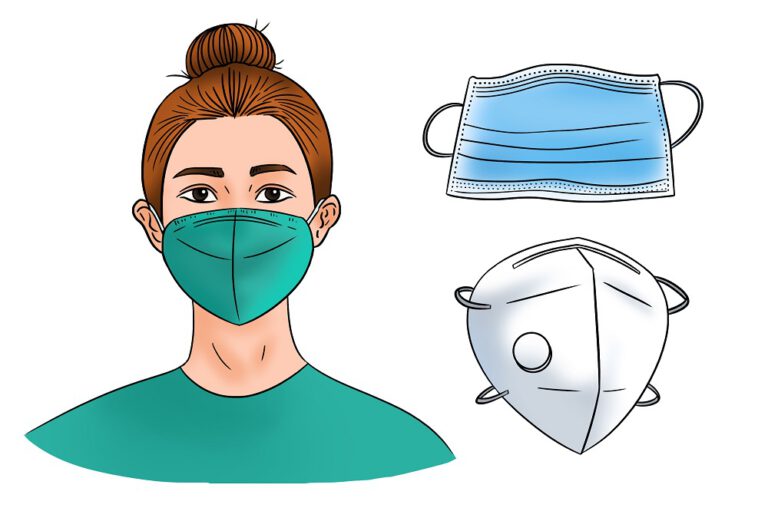
How Your Mask Protects You(Up date)
Correct and consistent mask use is a critical step everyone can take to prevent getting and spreading COVID-19. Masks work best when everyone wears them, but not all masks provide the same protection. When choosing a mask, look at how well it fits, how well it filters the air, and how many layers it has.
Ensuring the Safety of COVID-19 Vaccines
The United States currently has the safest vaccine supply in its history. Vaccine Safety is a vital part of the nation’s response to the COVID-19 pandemic. As vaccines are developed and become available, the public’s knowledge of their safety, both initially and during extended use, is an important part of a successful vaccination effort.

Benefits of Getting a COVID-19 Vaccine
The Corona vaccine teaches our immune system how to recognize and fight the virus that causes COVID-19. Sometimes this process can cause symptoms, such as fever. These symptoms are normal and are a sign that the body is building protection against the virus that causes COVID-19. So vaccines are needed to protect us…
Let’s get acquainted with the New Variant of Corona virus
A new variant strain of SARS-CoV-2 that contains a series of mutations has been described in the United Kingdom (UK) and become highly prevalent in
New Variant of Virus that Causes COVID-19 Detected
A new variant strain of SARS-CoV-2 that contains a series of mutations has been described in the United Kingdom (UK) and become highly prevalent in London and southeast England. Based on these mutations, this variant strain has been predicted to potentially be more rapidly transmissible than other circulating strains of SARS-CoV-2.
Corona virus vaccine made in Iran
Any drug or vaccine that is mass-produced must first go through a stage in the laboratory, and after the initial stages are completed, the test is performed on animals, and after receiving a positive result, further research is performed on a small number of humans.
“Who should not get the Pfizer vaccine?”
People with a history of severe allergic reactions should not get the Pfizer / Bioanth vaccine. This recommendation includes carriers of people who have been allergic to drugs, food or vaccines in the past. Those who have been shown to be allergic to the Pfizer vaccine have been treated and recovered. Allergic reactions are usually rare, but can occur with certain vaccines, such as the annual flu shot.
Trust in the corona vaccine is the biggest global health challenge
The seeds of doubt are being sown before a vaccine has even been approved. It is time to prepare the public for what could be a significant hurdle for any COVID-19 vaccine.

Covid-19 virus rapid antigen detection kits
The gold standard and definitive reference method for corona diagnosis is PCR tests, which due to the high sensitivity of this test requires a lot of precision and must be performed by a skilled technician in this process. As the pandemic prolonged and the incidence of the disease increased, the high cost of the test and, most importantly, the length of time it took to perform the test forced researchers to use simpler diagnostic methods that could be performed in a short time.
Comparison of “Moderna” and “Pfizer” vaccines
The close competition to create a vaccine for COVID-19 by the world’s leading biopharmaceutical companies has given people a fresh hope that the biggest pandemic of the century will be eventually eradicated. Meanwhile Pfizer and Moderna have recently announced their COVID-19 vaccines are more than 90% effective.

Introduction to the method of making Corona virus vaccine by two big companies “BIONTECH” and “PFIZER”R”
today we have a new update about development of Covid-19 vaccines. scientific minds around the world look to provide an end to the current coronavirus pandemic. We are waiting for FDA approval for pizer and biontech vaccine
Introduction to the method of making Corona virus vaccine by two big companies “Oxford” and “Moderna”
Updates around the development of Covid-19 vaccines seems to be an almost daily occurrence at the moment, as the best medical and scientific minds around the world look to provide an end to the current coronavirus pandemic.
Vaccines from Moderna have produced high efficacy ratings in late trials, Janssen has launched a late stage clinical trial and now the University of Oxford / AstraZeneca jab has also returned encouraging results.
Steps To Make a Corona Vaccine
Throughout this pandemic, you’ve likely heard terms like Phase 1-2a trial and FDA approval, but what do these really mean—and at what point do they happen?
A vaccine would allow the body to safely develop an immune response to COVID-19 that could prevent or control infection.
it takes time to develop safe and effective vaccines – usually five to ten years on average. Despite promising reports about potential coronavirus vaccines being developed worldwide, it could take an estimated 12-18 months to develop one.
How Vaccines Work
Vaccines contain the same germs that cause disease. (For example, measles vaccine contains measles virus, and Hib vaccine contains Hib bacteria.) But they have been either killed or weakened to the point that they don’t make you sick. Some vaccines contain only a part of the disease germ.
Corona virus and blood types
Blood type seems to be a predictor of how susceptible a person is to contracting SARS-CoV-2, though scientists haven’t found a link between blood type per se and severity of disease.

Criteria for Return to Work – healthcare personnel
Occupational health programs and public health officials making decisions about return to work for healthcare personnel (HCP) with confirmed SARS-CoV-2 infection, or who have suspected SARS-CoV-2 infection (e.g., developed symptoms of COVID-19) but were never tested for SARS-CoV-2.

When can I be around others?
I think or know I had COVID-19, and I had symptoms You can be around others after

Coronavirus disease (COVID-19) advice for the public
Most countries around the world have seen cases of COVID-19 and many are experiencing outbreaks. Authorities in China and some other countries have succeeded in slowing their outbreaks. However, the situation is unpredictable so check regularly for the latest news.
COVID-19 and Seasonal Allergies
An allergy (allergic rhinitis) that occurs in a particular season is more commonly known as hay fever . About 8 percent of Americans experience it. Hay

Coronavirus and Tobacco
Tobacco consumption, specially smoking and waterpipe, affect the lung capacity which may lead to difficulty in breathing, caused by long term harmful effect on cardiovascular and respiratory system leading to elevated risk of various kind of infectious diseases.

Do’s and Don’ts of Using Masks
the optimal way to prevent airborne transmission is to use a combination of interventions from across the hierarchy of controls, not just PPE alone.

COVID-19 recovery at home: Information for patients and their families
You have confirmed (or possible) COVID-19, and you are being sent home to rest and recover, as your symptoms do not require hospitalization at this time
Brain damage and coronavirus
More than Six months into the COVID-19 pandemic, we’re still learning what the disease can do.One key thing we’re seeing is that severity of lung illness doesn’t always correlate with severity of neurological illness. Having only minor lung illness doesn’t protect against potentially severe complications.

What do you know about the flu virus and its vaccine?
Flu is a contagious respiratory illness caused by influenza viruses that infect the nose, throat, and sometimes the lungs. It can cause mild to severe illness, and at times can lead to death. The best way to prevent flu is by getting a flu vaccine each year
Similarities and differences between Covid-19 and the flu
As the COVID-19 outbreak continues to evolve, comparisons have been drawn to influenza. Both cause respiratory disease, yet there are important differences between the two viruses and how they spread. This has important implications for the public health measures that can be implemented to respond to each virus


